Fresh osteochondral graft. Indications, surgical technique and scientific evidence
Injerto de cartílago en fresco. Indicaciones, técnica quirúrgica y evidencia científica
Resumen:
Las lesiones condrales de la rodilla de espesor completo suponen un desafío para el cirujano ortopédico cuando se presentan en pacientes jóvenes y son sintomáticas. El aloinjerto osteocondral en fresco es un procedimiento válido para tratar grandes defectos osteocondrales de rodilla en pacientes jóvenes y activos, en los que la cirugía protésica no está indicada. Esta técnica quirúrgica ofrece la posibilidad de restaurar de manera inmediata la estructura histológica condral y ósea original sin añadir comorbilidad a la zona donante. Puede ser también utilizada en casos complejos de reconstrucción articular en el contexto de osteonecrosis, osteocondritis disecante, mala unión, artritis postraumática o en procedimientos de revisión. Los bancos de células y tejidos han mejorado la disponibilidad de este tipo de aloinjerto gracias a la optimización de las técnicas de procesado y de almacenamiento. Asimismo, la aplicación de protocolos estrictos de cribado de los donantes mediante pruebas serológicas aseguran un riesgo de transmisión de enfermedades mínimo.
Para asegurar la supervivencia del injerto, es necesario realizar una adecuada selección del paciente, así como evaluar las condiciones de malalineación, estabilidad ligamentosa y déficit meniscal. El objetivo de este trabajo es revisar las indicaciones y los aspectos más destacados de la técnica quirúrgica, además de describir la tasa de supervivencia del aloinjerto osteocondral en fresco en distintas localizaciones de la rodilla.
Abstract:
Full-thickness chondral lesions of the knee are a challenge for orthopedic surgeons when manifesting with symptoms in young patients. Fresh osteochondral allografts are a valid option for treating large osteochondral defects of the knee in young and active individuals in which prosthetic surgery is not indicated. This surgical technique allows immediate restoration of the original bone and chondral histological structure without adding comorbidity to the donor zone. It can also be used in complex joint reconstruction cases in the context of osteonecrosis, osteochondritis dissecans, malunion, post-traumatic osteoarthritis, or in revision surgery. Cell and tissue banks have improved the availability of this type of allograft thanks to optimization of the processing and storage techniques. Likewise, the application of strict donor screening protocols based on serological tests ensures a minimum risk of disease transmission.
In order to ensure graft survival, adequate patient selection is required, with the evaluation of misalignment, ligament stability and meniscus deficit. The purpose of the present study was to review the indications and main aspects of the surgical technique, and to describe the fresh osteochondral allograft survival rate in different locations of the knee.
Background
Knee joint cartilage injuries are frequent in young and active people (between 26-47 years of age)(1). The prevalence of these lesions in arthroscopic evaluation is approximately 65%, and of these, 5-20% are high grade lesions(2,3,4,5,6). In most cases, joint cartilage damage is characterized by insidious pain and knee effusion in relation to sports activity, and has a strong impact upon quality of life - not only during sports but also in activities of daily living(7). In patients with full-thickness chondral lesions, chondral repair techniques may prove necessary in up to 11% of all cases(5).
In patients of this kind, replacement arthroplasty has poor satisfaction outcomes and moreover entails a high risk of revision surgery due to loosening or wear(8,9). The aim of surgery therefore should be to restore joint biology, alleviate pain, improve function and delay or eliminate the need for prosthetic surgery.
At present, the use of autografts (mosaicplasty) or fresh allografts offers the possibility of immediate restoration of the original chondral histological structure. However, autografts have the inconvenience of damaging the healthy zone from which the graft is harvested. In addition, they do not have the same form, density and thickness of the chondral tissue, since they come from zones different from the recipient zone.
Allogenic transplantation allows replacement of the damaged joint cartilage and subchondral bone with mature hyaline cartilage containing viable chondrocytes from the same anatomical region, along with the underlying healthy bone. It also allows us to cover virtually all types of defects and restore complex surfaces and non-contained lesions(10,11,12,13,14,15,16,17,18,19). Furthermore, the technique does not cause donor zone morbidity and induces only a minimum immune response in the recipient, without clinical repercussions(20). In this way, fresh osteochondral allografting is a valid indication for large osteochondral defects of the knee(17,21). This is particularly important in young patients in which previous surgeries have failed.
However, the use of fresh osteochondral allografts poses logistic and medical problems. In our setting, the obtainment of an adequate graft, in both cartilage quality and morphological terms, constitutes one of the main limitations of this surgical technique. Another problem is the programming of surgery; the viability period from donor obtainment is 2-4 weeks, depending on the processing method involved. On the other hand, the time delay in obtaining the quality control results (normally microbiological controls) of the graft is one week. This means that the margin available for performing surgery is 1-3 weeks. Other concerns apart from cellular viability of the cartilage are graft immunogenicity and the risk of infectious disease transmission.
The presence of viable chondrocytes is crucial, as they are essential for maintenance of the extracellular matrix and for preventing graft degeneration over time. In fact, there is a direct correlation between chondrocyte viability and the success of osteochondral transplantation(19).
Cell and tissue banks process and store therapeutic resources for tissue repair, with the ethical need to offer patients products that are safe from the biological perspective, and clinically effective.
Fundamentally, these banks have two allograft storage options: fresh frozen and cryopreservation. The former is the most widely used option for tissue preservation in our setting, but is associated to loss of cell life, which in turn results in chondral tissue preservation failure. On the other hand, chondral tissue cryopreservation has not been found to be effective. The presence of a dense and consistent extracellular matrix and the lack of vascularization complicate penetration of the cryoprotective substances. As a result, the cell viability (except in the case of small fragments measuring only a few mm3) obtained after thawing is not enough to guarantee the clinical efficacy of the allograft.
Another option is fresh preservation. Precisely the two characteristics that complicate satisfactory cryopreservation (avascularity and consistency of the matrix) here appear as modulators or limiting factors against an eventual immune response of the recipient. Accordingly, storage is proposed under conditions that allow availability of the allograft (with sufficient cell viability) long enough to perform the required quality controls (fundamentally intended to avoid the risk of disease transmission), and to program surgery. Tissue banks must validate the maximum fresh preservation time to guarantee the cell viability needed for implant success.
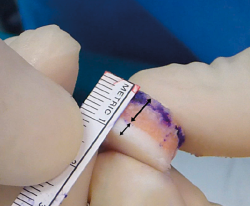
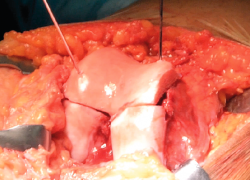
Since the graft is not vascularized, the osteocytes of the bone component of the allograft do not survive. However, the bone tissue remains structurally intact and is mechanically resistant - thus acting as a support for the hyaline cartilage and a binding link with the host or recipient bone. Over time, it is replaced by the recipient bone through a phenomenon known as creeping substitution(22,23). When using allografts, and considering that there is usually little subchondral bone involvement in acute lesions (except in high-energy impact scenarios), it is advisable to minimize the bone fraction of the tissue (6-8 mm in thickness) (Figure 1). This reduces the time required to replace the donor bone tissue with the recipient bone, with full incorporation of the bone graft. In any case, since nutrients reach the joint cartilage via diffusion through the synovial fluid and also from the underlying cancellous bone, it is necessary to guarantee conditions that facilitate consolidation of the bone fraction of the graft. In a baboon animal model, Malinin and Ouellete(24) observed the development of canals between the deepest cartilage layer and the subchondral bone in autografts - this suggesting that routes are developed over the clinical course to allow nutrients to reach the graft.
From the immunological perspective, it is not clear whether there is an immune response conditioning evolution of the graft. The studies made in this regard have evidenced the presence of antibodies targeted to collagen(25), as well as class I and class II antibodies(26), following the transplantation of cryopreserved osteochondral tissue. However, there are no data unequivocally identifying the generated antibodies as being specifically targeted to donor HLA (human leukocyte antigen), and information is also needed about the response to fresh osteochondral tissue transplantation. During surgery, and in order to reduce the presence of marrow stroma and hence its potential antigenic load, thorough bone washing under pressure is performed(27).
With respect to the storage of viable osteochondral tissue, although most banks that offer this service use a temperature of 4ºC, data from animal models have shown that cell viability can be optimized by "culturing" the tissue at 37ºC, and the effective storage time is moreover prolonged as a result (28,29).
In order to limit the risk of infectious disease transmission as far as possible, the hospital transplant coordination teams in Spain (which are responsible for donor selection) receive extensive training, and the tissue banks moreover follow strict donor screening protocols based on serological tests and aseptic processing of the samples. In addition, tissue manipulation in the bank is regulated to minimize the risk of environmental contamination (30,31). At present, thanks to the introduction of enzyme immunoassay (EIA) techniques combined with the development of chemiluminescent microparticle immunoassay (CMIA) and microparticle enzyme immunoassay (MEIA) and nucleic acid testing (NAT), the risk of disease transmission is extremely low.
Based on the above, banks must work to implement protocols seeking to optimize their care services: 1) development of methods to expand the therapeutic repertoire, incorporating composite joint tissue allografts; 2) optimization of storage conditions to ensure improved preservation of cell viability; 3) adaptation of incubation temperature to allow prolonged preservation of cell viability, maintaining the properties of the extracellular matrix; 4) development of specific culture media; and 5) elevation of hydrostatic pressure. In this way, culture medium nutrient diffusion is facilitated, and at the same time simulation of the biomechanical effect of movement is sought, since it also plays an important role in cell viability and function(32).
Indications and contraindications
Indications
- Young patients (under 50 years of age) not amenable to prosthetic replacement surgery.
- Localized unipolar or bipolar grade III and IV lesions.
- Primary management of chondral or osteochondral lesions over 2 cm in size, independently of whether they are contained or not.
- Osteochondritis dissecans (OCD).
- Spontaneous osteonecrosis of the knee.
- Large post-traumatic osteochondral lesions.
- Failed chondral repair revision surgery.
Contraindications
- Body mass index (BMI) > 30 kg/m2.
- Tricompartmental degenerative conditions.
- Systemic inflammatory diseases, presence of infection or a history of osteomyelitic infection in the graft receptor zone, and active neoplastic disease.
- Medical conditions that may affect graft consolidation, such as insulin-dependent diabetes or smoking.
Technical aspects
When considering the transplantation of a fresh osteochondral allograft, we first must select a donor specimen of a size, side and form similar to those of the affected receptor area. This is particularly important in the case of the femoropatellar joint and in the tibial plateau. In this regard we can use anteroposterior and lateral projection radiographs under loading and Merchant view(33). Preoperative computed tomography is more precise, and is moreover useful for assessing subchondral bone involvement. Once the measures of the required graft have been obtained, they are forwarded to the tissue bank to thus locate the ideal donor. Likewise, the tissue bank must offer detailed information about the dimensions of the allograft (if possible, performing a computed tomography scan of the latter) to allow its suitability to be confirmed (particularly in cases of patellar or femoral trochlear involvement).
As with other chondral repair procedures, it is important to know the axial and coronal misalignment conditions, meniscal absence or ligament instability. These factors must be adequately evaluated and treated. The same surgical step can be used for fresh osteochondral allograft transplantation with ligament reconstruction, meniscus transplant and/or corrective osteotomies(34).
Meniscus damage is a relative contraindication or at least a factor to be resolved on an associated basis, since the meniscus plays a fundamental role in the absorption and distribution of loads over the joint surface. We therefore must repair the peripheral lesions and, in the case of considerable deficiencies, a meniscus transplant should be made to improve survival of the osteochondral allograft.
Misalignment is to be assessed from radiographs of the extremity under loading conditions. If the mechanical axis passes through the affected compartment, a corrective osteotomy should be added to unload and protect the allograft. Special care is required in the case of decompressive osteotomies, since deviations of as little as 2-3º may deserve correction during the same surgical procedure. In general, opening osteotomies are preferred when plug or dowel allograft techniques are performed. In contrast, closing osteotomies are reserved for massive or shell allografts. As far as possible, it is advisable to perform the osteotomy in bone of the joint opposite to the lesion in order to minimize surgical procedures in the same bone (medial distal femoral closing osteotomy and osteochondral allograft of the lateral plateau for external plateau osteochondral lesions associated to marked genu valgum).
When a femoropatellar fresh osteochondral allograft transplant is performed, maltracking must be discarded, since it may be an underlying cause of allograft failure. In these cases we can associate techniques targeted to soft tissues, such as release or elongation of the lateral retinaculum, reconstruction of the medial femoropatellar ligament, trochleoplasties or bone realignments(35).
Rotational and translational instability can damage the graft due to the shear forces it exerts upon the latter. In such cases ligament repair/reconstruction techniques are indicated.
Ideally, surgery should be performed by two surgical teams working simultaneously. One team prepares the graft and the other performs arthrotomy, preparation of the receptor zone, and graft implantation. Intravenous antibiotic prophylaxis is provided according to the usual protocol, and general anesthesia or spinal anesthesia is administered. The patient is placed in supine decubitus on the surgical table, with the ischemia cuff on the proximal thigh.
Before arthrotomy, diagnostic arthroscopy better characterizes the lesion and ensures a correct surgical approach. The options for access to the damaged zone depend on the affected area and the preferences of the surgeon. The more commonly used straight incision over the midline, followed by access to the joint through a medial or lateral parapatellar arthrotomy, can be alternated with a parapatellar incision and lateral arthrotomy if the lesion only affects the trochlea or patella. In patients with femorotibial lesions, adaptation to each case is logically required - the most common practice being the use of anterolateral or anteromedial longitudinal incisions, depending on the case. When treating patellar lesions, it is advisable not to remove Hoffa's fat pad in excess, in order to avoid patellar devascularization. Likewise, proximal, medial and lateral denervation of the mentioned bone is indicated.
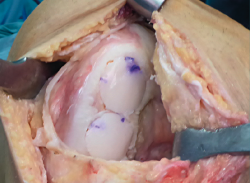
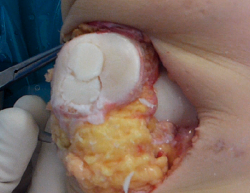
Dowel or plug, or shell allograft techniques are the most widely used procedures. In the case of solitary, well delimited and easily accessible defects, such as in the medial femoral condyle, use is made of plug allografts. The diameter of the graft can be precisely adjusted to the size of the lesion, affording complete coverage of the affected area and stable integration with the recipient bone and surrounding cartilage surfaces. In large or multiple lesions where a plug is unable to cover the entire defect - particularly if the length of the lesion is greater than its width - we can use a "snowman" (Figure 2) or "mastercard" (Figure 3) plug configuration, though this has been associated to increased revision surgery rates(36).
In the case of asymmetrical lesions (complete patella or extensive lesions of the trochlea), or in locations that prove difficult to access, use is made of the shell technique.
Femoral plug allografts
After exposing the lesion, the walls of healthy cartilage must be identified. Then, the size of the lesion is determined using the measurement templates that come with the allograft transplantation instruments.
After determining the size, a Kirschner needle is placed at the center of the lesion and perpendicular to its surface. A circular drill of the chosen size is used to work in-depth approximately 6-8 mm, without exceeding 10 mm. If the depth of the subchondral lesion, after debridement down to healthy bone, is over 8-10 mm, filling with autologous bone graft material (iliac crest or proximal tibia) should be carried out until achieving an ideal depth of 6-8 mm. With the prepared receptor bed, we measure the depth in the 12, 3, 6 and 9 o'clock positions.
On the graft preparation table, a trephine of the same size is used to extract a plug from the fresh allograft provided by the tissue bank. The excess subchondral bone is withdrawn and the 12 o'clock position is marked for correct orientation. In order to eliminate the remaining subchondral bone marrow, we irrigate (as a pulsatile flow) at least 6 litres of physiological saline solution. This step is made with each of the pieces before graft implantation in the receptor zone.
The surgeon in charge of preparing the allograft then transfers it to the lesion site. If the graft accidentally falls to the floor, a new washing step is advised, lasting at least 20 minutes, with immersion in a 1 g/100 ml vancomycin solution for 10 minutes.
The graft is placed with gentle pressure from the thumb or an appropriate impactor. Fixation is normally not needed, but reabsorbable screws or pins can be used if so required. The knee is mobilized over its full mobility range to assess stability of the graft or impingement zones.
Femoral shell allografts
After identifying the condylar lesion, the receptor zone is prepared. It should have a depth of 4-5 mm and a flat surface. The necrotic or damaged bone tissue must be removed.
The graft is traced on a paper template slightly oversized with respect to the receptor area. Gradual manual adjustment is made using a rongeur until the definitive size is obtained. Temporary fixation with Kirschner needles is made, and possible impingement zones along the full mobility range are discarded. Definitive fixation is carried out using reabsorbable pins or compression screws.
Patellar allografts
The shell technique is indicated in patients with extensive patellar osteochondral lesions. The surgical technique is the same as that used in prosthetic surgery when placing a patellar implant. After measuring the patellar thickness and surface, a sectioning plane is established parallel to that passing through the patellar and quadricipital tendons, using the knee prosthesis instruments and an oscillating saw piece. In order to minimize fracture risk, a thickness of between 12-15 should be maintained. The patella is systematically denervated through electrocoagulation, with the theoretical aim of reducing the incidence of postoperative anterior knee pain.
The allograft is prepared likewise using the knee prosthesis instruments; its thickness will be that affording a patellar thickness identical to that of the original, once implanted. If the graft is a little larger (as sometimes happens), it should be trimmed to adapt it to receptor size. The proximal and lateral part of the implant is marked so that the position of the graft is adequate when transferred to the receptor zone.
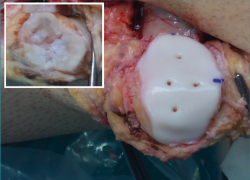
Provisional fixation is carried out with two Kirschner needles inserted through the anterior cortical layer of the patella, and joint congruence and tracking are checked. Definitive fixation in turn is made with 3-4 compression screws from the dorsal surface of the patella, or preferably using reabsorbable pins from the joint surface (Figure 4), allowing postoperative assessment of the osteochondral tissue based on magnetic resonance imaging (MRI)(23).
Tibial plateau allografts
Tibial plateau allografts are usually indicated in patients presenting sequelae of medial or lateral tibial plateau fractures.
An oscillating saw is used to cut 1-2 cm in depth within the plateau, according to the size of the defect. The cut should be perpendicular to the tibial axis, maintaining the posterior slope and preserving coronal and sagittal alignment. We preserve the tibial eminence or spine and the root of the contralateral meniscus in the sagittal cut. The meniscus is removed from the affected compartment, preserving the wall where the meniscal allograft will be sutured.
After preparing the size and shape of the tibial osteochondral allograft, temporary fixation with Kirschner needles is made, and stability over the full mobility range is assessed. Definitive fixation is achieved with 2-3 cancellous bone screws measuring 3.5 mm in diameter, followed by meniscal allograft suturing over the residual meniscal wall(37).
Femoral trochlea allografts
As commented, the technique based on one or two plugs is used in the case of small defects. In the same way as with central patellar lesions, preparation of the receptor bed and extraction of the allograft plugs may be complicated by failure to achieve perfect alignment of the circular cut template in the central trochlear zones, due to the double joint contour.
Shell allografting is advised in the case of large lesions, bipolar defects or defects involving practically the entire trochlea, and especially in patients with dysplastic trochlear lesions. It is crucial to select a trochlear allograft of adequate size, receptor side and form. Resection of the receptor trochlea is carried out in a way similar to anterior cut of the distal femur performed when placing a knee prosthesis, or preferably using 3-4 Kirschner needles serving as guides oriented 45º with respect to the coronal plane - thus allowing us to more efficiently advance towards the intercondylar zone (Figure 5). Once the graft has been provisionally placed, and after checking joint congruence and tracking, fixation is carried out with two medial screws and two lateral screws, or using reabsorbable pins.
Results. Literature review. Scientific evidence
Although there is no evidence based on randomized controlled trials regarding fresh osteochondral allograft transplantation, many short-, medium- and long-term studies are available, since this technique has been used for almost 50 years. Many of the studies report high survival rates and good and/or excellent functional and patient satisfaction outcomes. In other patients, the incidence of recovery of activity levels equivalent to those before the lesion is close to 80%(38,39,40).
The largest published retrospective series (129 knees) of fresh osteochondral allograft transplantation reported a survival rate of 82% at 10 years, 74% at 15 years and 66% at 20 years(17). In the mentioned study, the identified graft failure risk factors were a patient age of over 30 years and a history of two or more previous surgeries. These survival outcomes are similar to those reported in other earlier series(16,41).
On the other hand, the series with the longest duration of follow-up to date(42) (mean 21.8 years) recorded a survival rate of 91% at 10 years, 84% at 15 years, 69% at 20 years and a predicted estimated rate of 59% at 25 years. In that study, 90% of the patients were under 40 years of age and presented a high physical activity demand.
In contrast, fewer published data are available on tibial plateau osteochondral lesions, since injuries in this location that are amenable to treatment with this technique are less common. Shasha et al.(43), with a mean follow-up period of 12 years, recorded a survival rate of 80% at 10 years and 65% at 15 years. These results are similar to those of Gross et al.(16), who moreover reported reconversion to prosthesis in one-third of the allografts, due to failure of the latter after approximately 10 years.
Spontaneous osteonecrosis of the knee is characterized by focal osteonecrosis of the medial femoral condyle of the knee that can progress towards severe osteoarthrosis of the medial femorotibial joint. It is different from osteonecrosis secondary to alcoholism, chronic corticosteroid use or systemic diseases. In most cases it responds to conservative management in its early stages. In contrast, advanced stage lesions or osteochondral defects larger than 40% of the condyle width have an unfavourable prognosis and evolve towards osteoarthrosis(44). Fresh osteochondral allograft transplantation has been used in large lesions and also after failed surgery(45). Tirico et al.(46) described 7 patients with stage 2 and 3 lesions of the medial femoral condyle, with a mean lesion size of 4.6 cm2 and a median percentage proportion between lesion size and condyle width of 56.8%. No graft failures were documented after 7 years of follow-up.
Osteochondritis dissecans that fails to improve with conservative treatment may be difficult to resolve from the clinical perspective, particularly in the case of mature skeletal structures and in patients with large and/or unstable osteochondral fragments. Although a number of options are available for treating chondral lesions, it is difficult to compare them, due to differences between the patient populations. It has been seen(47,48,49) that fresh osteochondral allograft transplantation in this disease affords good functional outcomes and high survival rates. The long-term survival (93% at 10 years) and functional outcomes are excellent despite the fact that this is a salvage technique and that the treated lesions involve the worst possible scenario (large size, stages II and III, and failed previous surgeries)(50). Therefore, although there is no consensus as to which treatment is best, fresh osteochondral allograft transplantation is a valid management option - with the added advantage of restoring both the chondral and the bone defect.
Bipolar chondral injuries of the femorotibial joint, whether associated to bone involvement or not, may be regarded as a relative contraindication, due to the limited success obtained of the clinical outcomes(51) and the considerable percentage of failures. This is more apparent when establishing comparisons with monopolar transplants in the femoral condyle(52,53,54,55). One of the causes of failure and thus of the poorer outcomes may be progression of the cartilage disease(56,57).
On the other hand, a relationship has been established between allograft size and transplant failure in injuries of this kind(58). Thus, it could be assumed that large bipolar osteochondral lesions may be indicative of advanced stages of chondral degeneration. This means that we must be more cautious when indicating fresh osteochondral allograft transplantation in large bipolar osteochondral defects. However, the relatively good clinical outcomes (both subjective and objective) recorded in patients with bipolar allografts that survive encourage the use of this technique when the patient age and activity level cause arthroplasty to be inadequate. Thus, although the outcomes are not as good as in the case of monopolar injuries, fresh osteochondral allograft transplantation can be regarded as a potential biological treatment option in patients with bipolar cartilage injuries of the knee.
Femoropatellar cartilage lesions pose a clinical management challenge, and as described in a recent systematic review (scientific level of evidence IV)(59): “it is not possible to define an ideal surgical procedure for lesions ≥ 4 cm2 in size in patients under 50 years of age”. In young patients, the implantation of femoropatellar prostheses should be limited, due to the risk of wear and loosening. If such patients present deep and extensive lesions, the use of massive fresh osteochondral allografts could be considered. However, few studies have been published on the use of massive fresh osteochondral allografts in application to the femoropatellar joint, and most of the existing information comes from case series(35,51,56,60,61).
Due to the anatomy of the trochlear sulcus, transplantation in this location is technically more demanding than in the tibiofemoral joint. In fact, the orientation of the graft in the coronal plane is more critical than in defects of similar size in the tibiofemoral joint(62). This may explain the allograft revision surgery rates reported in most of these series(35,51,56,60). Nevertheless, more recent studies describe very consistent long-term outcomes that are comparable to those of other chondral repair procedures, and should serve to encourage the use of allograft transplants for the femoropatellar joint. Gracitelli et al.(35) reported a survival rate of 78.1% at 10 years and of 55.8% at 15 years, while Cameron et al.(61) found survival to be 100% at 5 years and 91.7% at 10 years for isolated trochlear defects.
As commented, bipolar femorotibial lesions offer poorer outcomes, with an increased failure rate compared with monopolar defects. This does not seem to apply in the case of the femoropatellar joint, since considering the failure rate of monopolar allografts in this location and of bipolar allografts, the figures are similar(35,51,56,60). All this defines fresh osteochondral allograft transplantation as having an important role in osteochondral defects that are deep, bipolar and/or in revision procedures of the femoropatellar joint.
A key point for avoiding allograft failure is identification of the possible causes of axial misalignment, meniscal failure or ligament instability. It has been described that alignment of the knee and over 50% narrowing of the joint space are related to allograft failure(52). Likewise, grafts implanted in compartments subjected to greater compressive forces are histologically characterized by a delay in revascularization and creeping substitution, together with loss of joint cartilage(22). It is therefore essential to assess misalignment from the preoperative radiographs under loading conditions, in order to protect the allografts by means of a realignment osteotomy(43,63), even in the case of small angle deviations in the range of 2-3°. In fact, osteotomy concomitant to allografting affords better outcomes than when osteotomy is performed before or after grafting(16). Similar results have been reported in tibial plateau osteochondral allograft transplants that were associated to varizing osteotomies of the distal femur(64). On the other hand, although there may be a theoretical risk of altered vascular supply to the bone surrounding the graft, thereby affecting its integration, there are no data clearly corroborating this possibility(65).
Likewise, in the event of subluxation or asymmetrical loading of the lateral patellar facet, femoropatellar instability or incipient femoropatellar osteoarthritis, fresh osteochondral allograft transplantation should be combined with a medial patellofemoral ligament reconstruction procedure or femoropatellar realignment(56,62).
Conclusions
Fresh osteochondral allograft transplants are a useful option for treating large osteochondral defects of the knee in young and active individuals in which prosthetic replacement surgery is not expected to be a good solution. The current processing techniques ensure high chondrocyte viability, with a negligible risk of disease transmission. Adequate patient selection, as well as the assessment of misalignment, ligament stability and meniscal deficits are crucial for ensuring graft survival. Allografting of femoral defects affords excellent middle- and long-term outcomes, while the prognosis is adversely affected by chronic lesions, bipolar injuries, involvement of the femoropatellar joint, and older patient age.
Figuras
Figure 4. Definitive fixation of the patellar shell osteochondral allograft using reabsorbable pins.
Información del artículo
Cita bibliográfica
Autores
Alejandro Roselló-Añón
Servicio de Cirugía Ortopédica y Traumatología. Hospital Arnau de Vilanova. Valencia
Vicente Mirabet Lis
Banco de Células y Tejidos. Centro de Transfusión de Valencia
Óscar Fariñas Barbera
Vall d'Hebron Institut de Recerca (VHIR). Barcelona
Banc de Sang i Teixits. Instituto de Investigación Biomédica Sant Pau. Barcelona
Departamento de Anatomía y Embriología Humana Prof. D. Ruano Gil. Facultad de Medicina. Universidad de Barcelona
Pablo Eduardo Gelber Ghertner
Departamento de Cirugía Ortopédica. Hospital de la Santa Creu i Sant Pau. Universitat Autonoma de Barcelona (UAB). Barcelona
ReSport Clinic, Barcelona
Unidad de Artroscopia y Rodilla. ICATME-Institut Universitari Dexeus.
Vicente Sanchís Alfonso
Servicio de Traumatología y Cirugía Ortopédica. Hospital Arnau de Vilanova. Valencia
Ethical responsibilities
Conflicts of interest. The authors declare that they have no conflicts of interest.
Financial support. This study has received no financial support.
Protection of people and animals. The authors declare that this research has not involved studies in humans or in animals.
Data confidentiality. The authors declare that the protocols of their work centre referred to the publication of patient information have been followed.
Right to privacy and informed consent. The authors declare that no patient data appear in this article.
Referencias bibliográficas
-
1Flanigan DC, Harris JD, Trinh TQ, Siston RA, Brophy RH. Prevalence of chondral defects in athletes’ knees: a systematic review. Med Sci Sports Exerc. 2010;42(10):1795-801.
-
2Mankin HJ. The response of articular cartilage to mechanical injury. J Bone Joint Surg Am. 1982;64(3):460-6.
-
3Curl WW, Krome J, Gordon ES, Rushing J, Smith BP, Poehling GG. Cartilage injuries: a review of 31,516 knee arthroscopies. Arthroscopy. 1997;13(4):456-60.
-
4Hjelle K, Solheim E, Strand T, Muri R, Brittberg M. Articular cartilage defects in 1,000 knee arthroscopies. Arthroscopy. 2002;18(7):730-4.
-
5Arøen A, Løken S, Heir S, Alvik E, Ekeland A, Granlund OG, Engebretsen L. Articular cartilage lesions in 993 consecutive knee arthroscopies. Am J Sports Med. 2004 Jan-Feb;32(1):211-5.
-
6Widuchowski W, Widuchowski J, Trzaska T. Articular cartilage defects: study of 25,124 knee arthroscopies. Knee. 2007;14(3):177-82.
-
7Heir S, Nerhus TK, Røtterud JH, Løken S, Ekeland A, Engebretsen L, Arøen A. Focal cartilage defects in the knee impair quality of life as much as severe osteoarthritis: a comparison of knee injury and osteoarthritis outcome score in 4 patient categories scheduled for knee surgery. Am J Sports Med. 2010 Feb;38(2):231-7.
-
8Lonner JH, Hershman S, Mont M, Lotke PA. Total knee arthroplasty in patients 40 years of age and younger with osteoarthritis. Clin Orthop Relat Res. 2000;380:85-90.
-
9Bayliss LE, Culliford D, Monk AP, Glyn-Jones S, Prieto-Alhambra D, Judge A, et al. The effect of patient age at intervention on risk of implant revision after total replacement of the hip or knee: a population-based cohort study. Lancet. 2017 Apr 8;389(10077):1424-30.
-
10Assenmacher AT, Pareek A, Reardon PJ, Macalena JA, Stuart MJ, Krych AJ. Long-term outcomes after osteochondral allograft: a systematic review at long-term follow-up of 12.3 years. Arthroscopy. 2016;32(10):2160-8.
-
11Capeci CM, Turchiano M, Strauss EJ, Youm T. Osteochondral allografts: applications in treating articular cartilage defects in the knee. Bull Hosp Jt Dis (2013). 2013;71(1):60-7.
-
12Chahal J, Gross AE, Gross C, Mall N, Dwyer T, Chahal A, et al. Outcomes of osteochondral allograft transplantation in the knee. Arthroscopy. 2013;29(03):575-88.
-
13Chui K, Jeys L, Snow M. Knee salvage procedures: the indications, techniques and outcomes of large osteochondral allografts. World J Orthop. 2015;6(03):340-50.
-
14De Caro F, Bisicchia S, Amendola A, Ding L. Large fresh osteochondral allografts of the knee: a systematic clinical and basic science review of the literature. Arthroscopy. 2015;31(04):757-65.
-
15Görtz S, De Young AJ, Bugbee WD. Fresh osteochondral allografting for steroid-associated osteonecrosis of the femoral condyles. Clin Orthop Relat Res. 2010;468(05):1269-78.
-
16Gross AE, Shasha N, Aubin P. Long-term followup of the use of fresh osteochondral allografts for posttraumatic knee defects. Clin Orthop Relat Res. 2005;(435):79-87.
-
17Levy YD, Görtz S, Pulido PA, McCauley JC, Bugbee WD. Do fresh osteochondral allografts successfully treat femoral condyle lesions? Clin Orthop Relat Res. 2013;471(01):231-7.
-
18McCulloch PC, Kang RW, Sobhy MH, Hayden JK, Cole BJ. Prospective evaluation of prolonged fresh osteochondral allograft transplantation of the femoral condyle: minimum2-year followup. Am J Sports Med. 2007;35(03):411-20.
-
19Sherman SL, Garrity J, Bauer K, Cook J, Stannard J, Bugbee W. Fresh osteochondral allograft transplantation for the knee: current concepts. J Am Acad Orthop Surg. 2014;22(02):121-33.
-
20Hunt HE, Sadr K, Deyoung AJ, Gortz S, Bugbee WD. The role of immunologic response in fresh osteochondral allografting of the knee. Am J Sports Med. 2014;42(4):886-91.
-
21Williams RJ 3rd, Ranawat AS, Potter HG, Carter T, Warren RF. Fresh stored allografts for the treatment of osteochondral defects of the knee. J Bone Joint Surg Am. 2007;89(4):718-26.
-
22Oakeshott RD, Farine I, Pritzker KP, Langer F , Gross AE. A clinical and histologic analysis of failed fresh osteochondral allografts. Clin Orthop Relat Res. 1988;233:283-94.
-
23Gelber PE, Perelli S, Ibáñez M, Ramírez-Bermejo E, Fariñas O, Monllau JC, Sanchís-Alfonso V. Fresh Osteochondral Patellar Allograft Resurfacing. Arthrosc Tech. 2018;7(6):e617-e622.
-
24Malinin T, Ouellette EA. Articular cartilage nutrition is mediated by subchondral bone: a long-term autograft study in baboons. Osteoarthritis Cartilage. 2000;8(6):483-91.
-
25Bujia J, Alsalameh S, Naumann A, Wilmes E, Sittinger M, Burmester GR. Humoral immune response against minor collagens type IX and XI in patients with cartilage graft resorption after reconstructive surgery. Ann Rheum Dis. 1994;53:229-34.
-
26Friedlaender GE, Strong DM, Tomford WW, Mankin HJ. Long-term follow-up of patients with osteochondral allografts. A correlation between immunologic responses and clinical outcome. Orthop Clin North Am. 1999 Oct;30(4):583-8.
-
27Sun Y, Jiang W, Cory E, Caffrey JP, Hsu FH, Chen AC, et al. Pulsed lavage cleansing of osteochondral grafts depends on lavage duration, flow intensity, and graft storage condition. PLoS One. 2017;12(5):e0176934.
-
28Pallante AL, Bae WC, Chen AC, Görtz S, Bugbee WD, Sah RL. Chondrocyte viability is higher after prolonged storage at 37 degrees C than at 4 degrees C for osteochondral grafts. Am J Sports Med. 2009;37(Suppl 1):24S-32S.
-
29Garrity JT, Stoker AM, Sims HJ, Cook JL. Improved osteochondral allograft preservation using serum-free media at body temperature. Am J Sports Med. 2012 Nov;40(11):2542-8.
-
30Caselli-Fernández LM, Terkola R. Clean room environment, personnel, quality assurance and their monitoring. Eur J Hosp Pharm. 2006;12:29-34.
-
31Real Decreto-Ley 9/2014, por el que se establecen las normas de calidad y seguridad para la donación, la obtención, la evaluación, el procesamiento, la preservación, el almacenamiento y la distribución de células y tejidos humanos, y se aprueban las normas de coordinación y funcionamiento para su uso en humanos. BOE no. 163: 52716-63. Year 2014.
-
32Reinold MM, Wilk KE, Macrina LC, Dugas JR, Cain EL. Current concepts in the rehabilitation following articular cartilage repair procedures in the knee. J Orthop Sports Phys Ther. 2006;36:774-94.
-
33Pisanu G, Cottino U, Rosso F, Blonna D, Marmotti AG, Bertolo C, et al. Large Osteochondral Allografts of the Knee: Surgical Technique and Indications. Joints. 2018 Mar 13;6(1):42-53.
-
34Alford JW, Cole BJ. Cartilage restoration, Part 1: Basic science, historical perspective, patient evaluation, and treatment options. Am J Sports Med. 2005;33:295-306.
-
35Gracitelli GC, Meric G, Pulido PA, Görtz S, De Young AJ, Bugbee WD. Fresh osteochondral allograft transplantation for isolated patellar cartilage injury. Am J Sports Med. 2015;43(04):879-84.
-
36Familiari F, Cinque ME, Chahla J, Godin JA, Olesen ML, Moatshe G, LaPrade RF. Clinical Outcomes and Failure Rates of Osteochondral Allograft Transplantation in the Knee: A Systematic Review. Am J Sports Med. 2018 Dec;46(14):3541-9.
-
37Gelber PE, Erquicia J, Ramírez E, Fariñas O, Monllau JC. Fresh osteochondral and meniscus allografting for posttraumatic tibial plateau defects. Arthrosc Tech. 2018;7:e661-e667.
-
38Krych AJ, Pareek A, King AH, Johnson NR, Stuart MJ, Williams RJ III. Return to sport after the surgical management of articular cartilage lesions in the knee: a meta-analysis. Knee Surg Sports Traumatol Arthrosc. 2017;25(10):3186-96.
-
39Nielsen ES, McCauley JC, Pulido PA, Bugbee WD. Return to sport and recreational activity after osteochondral allograft transplantation in the knee. Am J Sports Med. 2017;45(07): 1608-14.
-
40McCarthy MA, Meyer MA, Weber AE, Levy DM, Tilton AK, Yanke AB, Cole BJ. Can Competitive Athletes Return to High-Level Play After Osteochondral Allograft Transplantation of the Knee? Arthroscopy. 2017 Sep;33(9):1712-7.
-
41Aubin PP, Cheah HK, Davis AM, Gross AE. Long-term followup of fresh femoral osteochondral allografts for posttraumatic knee defects. Clin Orthop Relat Res. 2001;(391 Suppl):S318-S327.
-
42Raz G, Safir OA, Backstein DJ, Lee PT, Gross AE. Distal Femoral Fresh Osteochondral Allografts: Follow-up at a Mean of Twenty-two Years. J Bone Joint Surg Am. 2014 Jul 2;96(13):1101-7.
-
43Shasha N, Krywulak S, Backstein D, Pressman A, Gross AE. Long-term follow-up of fresh tibial osteochondral allografts for failed tibial plateau fractures. J Bone Joint Surg Am. 2003;85-A(suppl 2):33-9.
-
44Aglietti P, Insall JN, Buzzi R, Deschamps G. Idiopathic osteonecrosis of the knee: aetiology, prognosis and treatment. J Bone Joint Surg Br. 1983;65(5):588-97.
-
45Bugbee W, Cavallo M, Giannini S. Osteochondral allograft transplantation in the knee. J Knee Surg. 2012;25(2):109-16.
-
46Tírico LEP, Early SA, McCauley JC, Bugbee WD. Fresh Osteochondral Allograft Transplantation for Spontaneous Osteonecrosis of the Knee: A Case Series. Orthop J Sports Med. 2017 Oct 3;5(10):2325967117730540.
-
47Garrett JC. Fresh osteochondral allografts for treatment of articular defects in osteochondrtitis dissecans of the lateral femoral condyle in adults. Clin Orthop Relat Res. 1994;(303):33-7.
-
48Emmerson BC, Gortz S, Jamali AA, Chung C, Amiel D, Bugbee WD. Fresh osteochondral allografting in the treatment of osteochondritis dissecans of the femoral condyle. Am J Sports Med. 2007;35(6):907-14.
-
49Lyon R, Nissen C, Liu XC, Curtin B. Can fresh osteochondral allografts restore function in juveniles with osteochondritis dissecans of the knee? Clin Orthop Relat Res. 2013;471(4):1166-73.
-
50Sadr KN, Pulido PA, McCauley JC, Bugbee WD. Osteochondral Allograft Transplantation in Patients With Osteochondritis Dissecans of the Knee. Am J Sports Med. 2016 Nov;44(11):2870-5.
-
51Chu CR, Convery FR, Akeson WH, Meyers M, Amiel D. Articular cartilage transplantation: clinical results in the knee. Clin Orthop Relat Res. 1999;360:159-68.
-
52Ghazavi MT, Pritzker KP, Davis AM, Gross AE. Fresh osteochondral allografts for post-traumatic osteochondral defects of the knee. J Bone Joint Surg Br. 1997 Nov;79(6):1008-13.
-
53Convery F, Akeson W, Meyers M. The operative technique of fresh osteochondral allografting of the knee. Opin Tech Orthop. 1997;7:340-4.
-
54Bugbee WD, Convery FR. Osteochondral allograft transplantation. Clin Sports Med. 1999 Jan;18(1):67-75.
-
55Bugbee WD. Fresh osteochondral allografts. J Knee Surg. 2002;15(3):191-5.
-
56Jamali AA, Emmerson BC, Chung C, Convery FR, Bugbee WD. Fresh osteochondral allografts: results in the patellofemoral joint. Clin Orthop Relat Res. 2005;437:176-85.
-
57Horton MT, Pulido PA, McCauley JC, Bugbee WD. Revision osteochondral allograft transplantations: do they work? Am J Sports Med. 2013 Nov;41(11):2507-11.
-
58Meric G, Gracitelli GC, Görtz S, De Young AJ, Bugbee WD. Fresh osteochondral allograft transplantation for bipolar reciprocal osteochondral lesions of the knee. Am J Sports Med. 2015 Mar;43(3):709-14.
-
59Noyes FR, Barber-Westin SD. Advanced patellofemoral cartilage lesions in patients younger than 50 years of age: is there an ideal operative option? Arthroscopy. 2013;29:1423-36.
-
60Torga Spak R, Teitge RA. Fresh osteochondral allografts for patellofemoral arthritis: long-term followup. Clin Orthop Relat Res. 2006;444:193-200.
-
61Cameron JI, Pulido PA, McCauley JC, Bugbee WD. Osteochondral allograft transplantation of the femoral trochlea. Am J Sports Med. 2016;44(03):633-8.
-
62Lattermann C, Kremser V, Altintas B. Use of Fresh Osteochondral Allografts in the Patellofemoral Joint. J Knee Surg. 2018;31(03):227-30.
-
63Gross AE, Aubin P, Cheah HK, Davis AM, Ghazavi MT. A fresh osteochondral allograft alternative. J Arthroplasty. 2002 Jun;17(4 Suppl 1):50-3.
-
64Drexler M, Gross A, Dwyer T, Safir O, Backstein D, Chaudhry H, et al. Distal femoral varus osteotomy combined with tibial plateau fresh osteochondral allograft for post-traumatic osteoarthritis of the knee. Knee Surg Sports TraumatolArthrosc. 2015;23(5):1317-23.
-
65Hsu AC, Tirico LEP, Lin AG, Pulido PA, Bugbee WD. Osteochondral Allograft Transplantation and Opening Wedge Tibial Osteotomy: Clinical Results of a Combined Single Procedure.Cartilage. 2018 Jul;9(3):248-254. doi: 10.1177/1947603517710307. Epub 2017 May 22.
Descargar artículo:
Licencia:
Este contenido es de acceso abierto (Open-Access) y se ha distribuido bajo los términos de la licencia Creative Commons CC BY-NC-ND (Reconocimiento-NoComercial-SinObraDerivada 4.0 Internacional) que permite usar, distribuir y reproducir en cualquier medio siempre que se citen a los autores y no se utilice para fines comerciales ni para hacer obras derivadas.
Comparte este contenido
En esta edición
- Update on the treatment of focal cartilage lesions of the knee
- Comparison of the intraarticular injection of platelet rich plasma (PRGF®) and hyaluronic acid (Hyalone®) in the treatment of chondral lesions: a randomized, prospective clinical study
- Microfractures or bone marrow stimulation (BMS): evolution of the technique
- Fresh osteochondral graft. Indications, surgical technique and scientific evidence
- Autologous matrix-induced chondrogenesis (AMIC)
- Instant CEMTRO Cell (ICC), high density autologous chondrocytes implantation
- Regeneration of joint cartilage: perspectives and future
- Autologous chondrocyte implant surgery of the knee
- Full thickness chondral ulcers of the patella and femoral condyle of the knee
Más en PUBMED
Más en Google Scholar
Más en ORCID


Revista Española de Artroscopia y Cirugía Articular está distribuida bajo una licencia de Creative Commons Reconocimiento-NoComercial-SinObraDerivada 4.0 Internacional.